

收稿日期: 2023-03-20
网络出版日期: 2023-08-17
基金资助
广西壮族自治区卫生健康委员会自筹经费科研课题(Z-A20221268);广西壮族自治区卫生健康委员会自筹经费科研课题(Z20201224)
Distribution of knockdown resistance genotypes in Aedes albopictus in Nanning, Guangxi Zhuang Autonomous Region, China, 2022
Received date: 2023-03-20
Online published: 2023-08-17
Supported by
Scientific Research Project of Health Commission of Guangxi Zhuang Autonomous Region(Z-A20221268);Scientific Research Project of Health Commission of Guangxi Zhuang Autonomous Region(Z20201224)
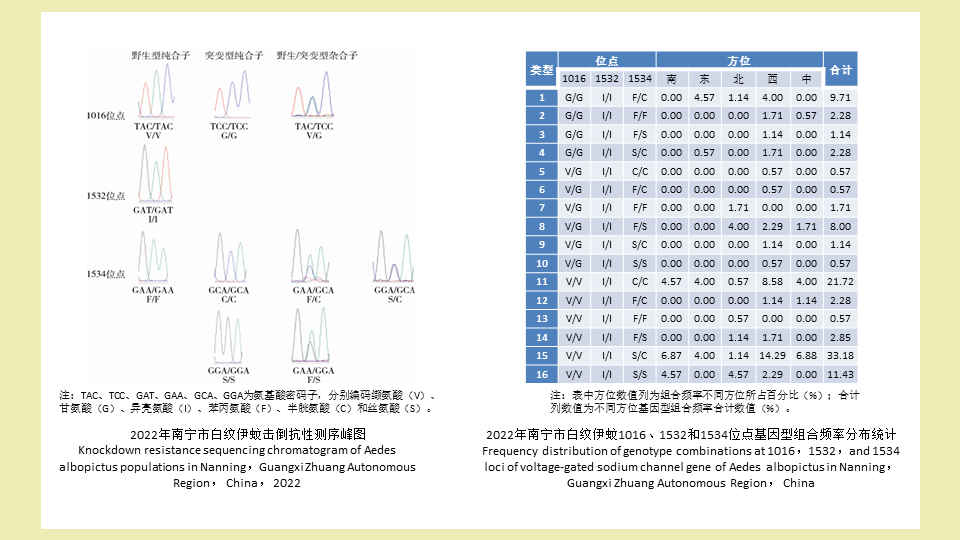
目的: 检测广西壮族自治区南宁市登革热媒介白纹伊蚊现场种群的击倒抗性基因型及其分布特点,以了解其抗药性水平,为科学防控白纹伊蚊提供依据。方法: 2022年在南宁市采用勺捕法采集伊蚊幼蚊,送实验室饲养至成虫,经形态学方法鉴定后,将白纹伊蚊用75%乙醇溶液浸泡,于-20 ℃保存备用。采用磁珠法试剂盒提取单只成蚊DNA,PCR扩增电压门控钠离子通道(VGSC)基因部分片段,测序结果在美国国立生物技术信息中心进行BLAST比对,对确认为白纹伊蚊VGSC基因的序列使用DNAStar 7.1分析其VGSC基因单位点及多位点联合突变情况。结果: 共检测175只白纹伊蚊,获得350条基因序列,长度约为400 bp,VGSC基因1532位点未发现突变,1016和1534位点均存在突变。1016位点有2种等位基因,分别为野生型1016V(148,78.28%)和突变型1016G(49,21.72%);3种基因型分别为野生型纯合子1016V/V(126,72.00%)、突变型纯合子1016G/G(27,15.43%)和野生/突变型杂合子1016V/G(22,12.57%)。1532位点有1种等位基因,即野生型1532I(175,100%);基因型均为野生型纯合子1532I/I。1534位点有3种等位基因,分别为野生型1534F(51,16.86%)、突变型1534S(116,36.29%)和突变型1534C(135,46.85%);6种基因型分别为野生型纯合子1534F/F(8,4.57%)、野生/突变型杂合子1534F/S(21,12.00%)、野生/突变型杂合子1534F/C(22,12.57%)、突变型杂合子1534S/C(64,36.57%)、突变型纯合子1534S/S(21,12.00%)和突变型纯合子1534C/C(39,22.29%)。结论: 南宁市VGSC基因突变率较高,应密切关注白纹伊蚊抗药性水平,科学、规范、高效地指导卫生杀虫剂的使用。
李雪, 凌峰, 韦舒琳, 屈志强, 刁书琴, 黄燕翠, 罗密芳 . 南宁市2022年白纹伊蚊击倒抗性基因型分布研究[J]. 中国媒介生物学及控制杂志, 2023 , 34(4) : 480 -484 . DOI: 10.11853/j.issn.1003.8280.2023.04.007
Objective: To investigate knockdown resistance (kdr) genotypes of Aedes albopictus which is the major vector of dengue in Nanning, so as to understand their insecticide resistance levels and provide evidence for scientific control of Ae. albopictus. Methods: Ae. albopictus larvae were collected using the dip method in Nanning and raised to adults in the laboratory. After morphological identification, Ae. albopictus mosquitoes were soaked in 75% ethanol and stored at -20 ℃. DNA was extracted from individual mosquitoes using the magnetic bead-based method. The partial fragments of the voltage-gated sodium channel (VGSC) gene were amplified by PCR and sequenced for alignment with the Basic Local Alignment Search Tool on the platform of National Center for Biotechnology Information. DNAStar 7.1 was used to analyze single-site and multiple-site mutations in the confirmed VGSC gene of Ae. albopictus mosquitoes. Results: A total of 175 Ae. albopictus mosquitoes were examined in 2022, obtaining 350 sequences (about 400 bp in length). Mutations were detected at the 1016 and 1534 loci but not at the 1532 locus of the VGSC gene. At the 1016 locus, there were two alleles (wild-type V [148, 78.28%] and mutant G [49, 21.72%]) and three genotypes (wild-type homozygote V/V [126, 72.00%], mutant homozygote G/G [27, 15.43%], and wild-type/mutant heterozygote V/G [22, 12.57%]). At the 1532 locus, there was one allele (wild-type I [175, 100%]) and one genotype (wild-type homozygote I/I [175, 100%]). At the 1534 locus, there were three alleles (wild-type F [51, 16.86%], mutant S [116, 36.29%], and mutant C [135, 46.85%]) and six genotypes (wild-type homozygote F/F [8, 4.57%], wild/mutant heterozygotes F/S [21, 12.00%] and F/C [22, 12.57%], mutant heterozygote S/C [64, 36.57%], and mutant homozygotes S/S [21, 12.00%] and C/C [39, 22.29%]). Conclusion: VGSC gene mutations are frequent in Ae. albopictus in Nanning. Attention should be paid to the insecticide resistance levels of Ae. albopictus for scientific, standardized, and efficient use of insecticides.

| 1 | 刘起勇. 我国登革热流行新趋势、防控挑战及策略分析[J]. 中国媒介生物学及控制杂志, 2020, 31 (1):1-6. |
| 1 | Liu QY. Dengue fever in China: New epidemical trend, challenges and strategies for prevention and control[J]. Chin J Vector Biol Control, 2020, 31 (1):1-6. |
| 2 | World Health Organization. Pesticides and their application, for the control of vectors and pests of public health importance, sixth edition [EB/OL]. (2006) [2023-03-02]. https://apps.who.int/iris/handle/10665/69223. |
| 3 | World Health Organization. Dengue guidelines for diagnosis, treatment, prevention and control, new edition [EB/OL]. (2009)[2023-03-02]. https://apps.who.int/iris/handle/10665/44188. |
| 4 | 赵春春, 周欣欣, 李文玉, 等. 2020年中国13省份登革热媒介白纹伊蚊抗药性监测及分析研究[J]. 中国媒介生物学及控制杂志, 2022, 33 (1):30-37. |
| 4 | Zhao CC, Zhou XX, Li WY, et al. Insecticide resistance surveillance and characteristic analysis of dengue vector Aedes albopictus in 13 provinces of China in 2020[J]. Chin J Vector Biol Control, 2022, 33 (1):30-37. |
| 5 | Hemingway J, Ranson H. Insecticide resistance in insect vectors of human disease[J]. Annu Rev Entomol, 2000, 45, 371-391. |
| 6 | Itokawa K, Sekizuka T, Maekawa Y, et al. High-throughput genotyping of a full voltage-gated sodium channel gene via genomic DNA using target capture sequencing and analytical pipeline MoNaS to discover novel insecticide resistance mutations[J]. PLoS Negl Trop Dis, 2019, 13 (11):e0007818. |
| 7 | Kasai S, Ng LC, Lam-Phua SG, et al. First detection of a putative knockdown resistance gene in major mosquito vector, Aedes albopictus[J]. Jpn J Infect Dis, 2011, 64 (3):217-221. |
| 8 | Kasai S, Caputo B, Tsunoda T, et al. First detection of a VSSC allele V1016G conferring a high level of insecticide resistance in Aedes albopictus collected from Europe (Italy) and Asia (Vietnam), 2016: A new emerging threat to controlling arboviral diseases[J]. Eurosurveillance, 2019, 24 (5):1700847. |
| 9 | Zhou XJ, Yang C, Liu N, et al. Knockdown resistance (kdr) mutations within seventeen field populations of Aedes albopictus from Beijing, China: First report of a novel V1016G mutation and evolutionary origins of kdr haplotypes[J]. Parasit Vectors, 2019, 12 (1):180. |
| 10 | 朱彩英, 赵春春, 伦辛畅, 等. 云南省景洪市2018-2019年白纹伊蚊击倒抗性基因型分布研究[J]. 中国媒介生物学及控制杂志, 2020, 31 (1):7-11. |
| 10 | Zhu CY, Zhao CC, Lun XC, et al. Distribution of knockdown resistance genotypes in Aedes albopictus in Jinghong, Yunnan province, China, 2018-2019[J]. Chin J Vector Biol Control, 2020, 31 (1):7-11. |
| 11 | 周小洁, 赵宇晗, 刘念, 等. 北京市两个白纹伊蚊种群kdr抗性突变检测分析[J]. 中华卫生杀虫药械, 2021, 27 (4):304-307. |
| 11 | Zhou XJ, Zhao YH, Liu N, et al. Detection and analysis of kdr resistance mutations in two populations of Aedes albopictus in Beijing[J]. Chin J Hyg Insect Equip, 2021, 27 (4):304-307. |
| 12 | 杨罗菊, 刘德星, 陈健, 等. 中山市白纹伊蚊现场种群击倒抗性基因检测分析[J]. 中国人兽共患病学报, 2021, 37 (2):171-175. |
| 12 | Yang LJ, Liu DX, Chen J, et al. Detection and analysis of the knockdown resistance gene in the field populations of Aedes albopictus in Zhongshan[J]. Chin J Zoonoses, 2021, 37 (2):171-175. |
| 13 | 赵春春. 我国白纹伊蚊抗药性及kdr基因分布研究[D]. 北京: 中国疾病预防控制中心, 2019. |
| 13 | Zhao CC. Study on insectcides resistance and kdr gene distribution of Aedes albopictus in China[D]. Beijing: Chinese Center for Disease Control and Prevention, 2019. (in Chinese) |
| 14 | 邱星辉. 白纹伊蚊抗药性分子机制研究进展[J]. 寄生虫与医学昆虫学报, 2019, 26 (3):194-198. |
| 14 | Qiu XH. Current knowledge about the molecular mechanisms underlying insecticide resistance in Aedes albopictus[J]. Acta Parasitol Med Entomol Sin, 2019, 26 (3):194-198. |
| 15 | 袁浩, 董昊炜, 庄松阳, 等. 电压门控钠离子通道基因I1532T突变与白纹伊蚊对菊酯类杀虫剂敏感性的相关性[J]. 中国热带医学, 2021, 21 (12):1130-1135, 1149. |
| 15 | Yuan H, Dong HW, Zhuang SY, et al. Correlation between the mutation of allele I1532T in voltage-gated sodium channel (VGSC) gene and susceptibility of Aedes albopictus to pyrethroid insecticides[J]. China Trop Med, 2021, 21 (12):1130-1135, 1149. |
| 16 | Auteri M, La Russa F, Blanda V, et al. Insecticide resistance associated with kdr mutations in Aedes albopictus: An update on worldwide evidences[J]. Biomed Res Int, 2018, 2018, 3098575. |
| 17 | Xu JB, Bonizzoni M, Zhong DB, et al. Multi-country survey revealed prevalent and novel F1534S mutation in voltage-gated sodium channel (VGSC) gene in Aedes albopictus[J]. PLoS Negl Trop Dis, 2016, 10 (5):e0004696. |
/
| 〈 |
|
〉 |