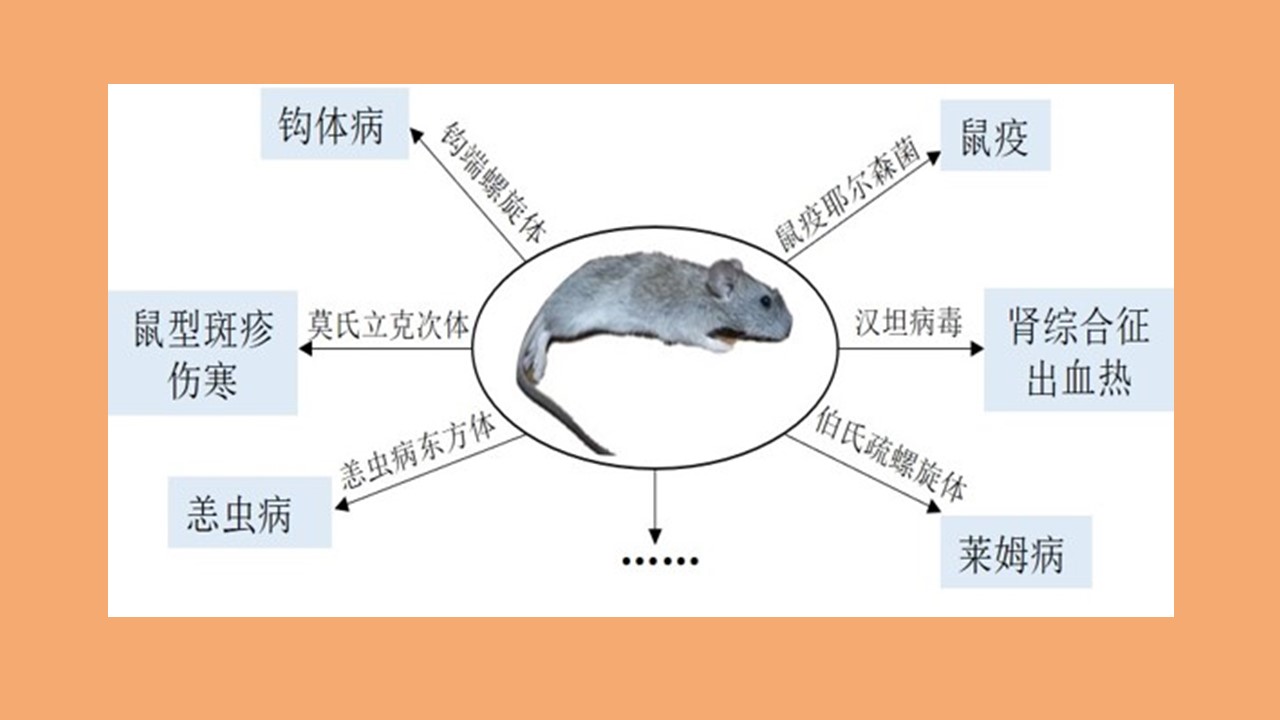
[1] Rabiee MH,Mahmoudi A,Siahsarvie R,et al. Rodent-borne diseases and their public health importance in Iran[J]. PLoS Negl Trop Dis,2018,12(4):e0006256. DOI:10.1371/journal.pntd.0006256.
[2] Griffiths J,Yeo HL,Yap G,et al. Survey of rodent-borne pathogens in Singapore reveals the circulation of Leptospira spp.,Seoul hantavirus,and Rickettsia typhi[J]. Sci Rep,2022,12(1):2692. DOI:10.1038/s41598-021-03954-w.
[3] 刘起勇.气候变化对中国媒介生物传染病的影响及应对:重大研究发现及未来研究建议[J].中国媒介生物学及控制杂志,2021,32(1):1-11. DOI:10.11853/j.issn.1003.8280.2021. 01.001. Liu QY. Impact of climate change on vector-borne diseases and related response strategies in China:Major research findings and recommendations for future research[J]. Chin J Vector Biol Control,2021,32(1):1-11. DOI:10.11853/j.issn.1003.8280. 2021.01.001.(in Chinese)
[4] Morris SR. Development of a recombinant vaccine against aerosolized plague[J]. Vaccine,2007,25(16):3115-3117. DOI:10.1016/j.vaccine.2007.01.071.
[5] 刘丽,汪巨峰,李波.鼠疫疫苗的研究现状和进展[J].中国药学杂志,2013,48(12):945-949. DOI:10.11669/cpj.2013.12.001. Liu L,Wang JF,Li B. Current status and progress of research on plague vaccine[J]. Chin Pharm J,2013,48(12):945-949. DOI:10.11669/cpj.2013.12.001.(in Chinese)
[6] Fitts EC,Andersson JA,Kirtley ML,et al. New insights into autoinducer-2 signaling as a virulence regulator in a mouse model of pneumonic plague[J]. mSphere,2016,1(6):e00342-16. DOI:10.1128/mSphere.00342-16.
[7] Zauberman A,Vagima Y,Tidhar A,et al. Host iron nutritional immunity induced by a live Yersinia pestis vaccine strain is associated with immediate protection against plague[J]. Front Cell Infect Microbiol,2017,7:277. DOI:10.3389/fcimb.2017. 00277.
[8] Demeure CE,Derbise A,Carniel E. Oral vaccination against plague using Yersinia pseudotuberculosis[J]. Chem Biol Interact,2017,267:89-95. DOI:10.1016/j.cbi.2016.03.030.
[9] Quenee LE,Ciletti NA,Elli D,et al. Prevention of pneumonic plague in mice,rats,guinea pigs and non-human primates with clinical grade rV10,rV10-2 or F1-V vaccines[J]. Vaccine,2011,29(38):6572-6583. DOI:10.1016/j.vaccine.2011.06.119.
[10] Hu JL,Jiao L,Hu YM,et al. One year immunogenicity and safety of subunit plague vaccine in Chinese healthy adults:An extended open-label study[J]. Hum Vaccin Immunother,2018,14(11):2701-2705. DOI:10.1080/21645515.2018.1486154.
[11] 李学荣,余新炳.核酸疫苗及其免疫机制研究[J].中国人兽共患病杂志,2000,16(6):82-86. DOI:10.3969/j.issn.1002-2694. 2000.06.029. Li XR,Yu XB. Research on nucleic acid vaccine and its immunization mechanism[J]. Chin J Zoonoses,2000,16(6):82-86. DOI:10.3969/j.issn.1002-2694.2000.06.029.(in Chinese)
[12] 焦磊,吴智远,王婷,等.鼠疫疫苗Ⅱa期临床血清对小鼠的被动保护水平及其与抗体效价的相关性分析[J].微生物学免疫学进展,2017,45(6):9-12. DOI:10.13309/j.cnki.pmi.2017. 06.002. Jiao L,Wu ZY,Wang T,et al. F1 and V antibody levels in correlation to mice protection passively adopted from human serum immunized by plague vaccine[J]. Prog Microbiol Immunol,2017,45(6):9-12. DOI:10.13309/j.cnki.pmi.2017. 06.002.(in Chinese)
[13] 陈化新.中国肾综合征出血热疫苗大规模应用研究[J].中华流行病学杂志,2002,23(2):145-147. DOI:10.3760/j.issn:0254-6450.2002.02.019. Chen HX. Large-scale application study of hemorrhagic fever vaccine for renal syndrome in China[J]. Chin J Epidemiol,2002,23(2):145-147. DOI:10.3760/j.issn:0254-6450.2002.02.019.(in Chinese)
[14] Sohn YM,Rho HO,Park MS,et al. Primary humoral immune responses to formalin inactivated hemorrhagic fever with renal syndrome vaccine (Hantavax®):Consideration of active immunization in South Korea[J]. Yonsei Med J,2001,42(3):278-284. DOI:10.3349/ymj.2001.42.3.278.
[15] 张锦鹏,张冠文,宣国云,等.溶酶体靶向的汉坦病毒包膜糖蛋白DNA疫苗的体液免疫评价和攻毒保护效果[J].中国免疫学杂志,2018,34(3):331-334. DOI:10.3969/j.issn.1000-484X.2018.03.003. Zhang JP,Zhang GW,Xuan GY,et al. Humoral immunity evaluation and virual challenge of lysosomal targeted hantavirus envelope glycoprotein DNA vaccine[J]. Chin J Immunol,2018,34(3):331-334. DOI:10.3969/j.issn.1000-484X.2018.03.003.(in Chinese)
[16] Brown KS,Safronetz D,Marzi A,et al. Vesicular stomatitis virus-based vaccine protects hamsters against lethal challenge with Andes virus[J]. J Virol,2011,85(23):12781-12791. DOI:10.1128/JVI.00794-11.
[17] Hooper J,Paolino KM,Mills K,et al. A phase 2a randomized,double-blind,dose-optimizing study to evaluate the immunogenicity and safety of a bivalent DNA vaccine for hemorrhagic fever with renal syndrome delivered by intramuscular electroporation[J]. Vaccines,2020,8(3):377. DOI:10.3390/vaccines8030377.
[18] Adler B. Vaccines against leptospirosis[J]. Curr Top Microbiol Immunol,2015,387:251-272. DOI:10.1007/978-3-662-45059-8_10.
[19] Adler B,de la Peña Moctezuma A. Leptospira and leptospirosis[J]. Vet Microbiol,2010,140(3/4):287-296. DOI:10.1016/j.vetmic.2009.03.012.
[20] Srikram A,Zhang KK,Bartpho T,et al. Cross-protective immunity against leptospirosis elicited by a live,attenuated lipopolysaccharide mutant[J]. J Infect Dis,2011,203(6):870-879. DOI:10.1093/infdis/jiq127.
[21] Conrad NL,McBride FWC,Souza JD,et al. LigB subunit vaccine confers sterile immunity against challenge in the hamster model of leptospirosis[J]. PLoS Negl Trop Dis,2017,11(3):e0005441. DOI:10.1371/journal.pntd.0005441.
[22] Forster KM,Hartwig DD,Seixas FK,et al. A conserved region of leptospiral immunoglobulin-like A and B proteins as a DNA vaccine elicits a prophylactic immune response against leptospirosis[J]. Clin Vaccine Immunol,2013,20(5):725-731. DOI:10.1128/CVI.00601-12.
[23] da Cunha CEP,Bettin EB,Bakry AFAAY,et al. Evaluation of different strategies to promote a protective immune response against leptospirosis using a recombinant LigA and LigB chimera[J]. Vaccine,2019,37(13):1844-1852. DOI:10.1016/j.vaccine. 2019.02.010.
[24] Garba B,Bahaman AR,Zakaria Z,et al. Antigenic potential of a recombinant polyvalent DNA vaccine against pathogenic leptospiral infection[J]. Microb Pathog,2018,124:136-144. DOI:10.1016/j.micpath.2018.08.028.
[25] Shen AK,Mead PS,Beard CB. The Lyme disease vaccine:A public health perspective[J]. Clin Infect Dis,2011,52 Suppl 3:S247-252. DOI:10.1093/cid/ciq115.
[26] Dattwyler RJ,Gomes-Solecki M. The year that shaped the outcome of the OspA vaccine for human Lyme disease[J]. npj Vaccines,2022,7(1):10. DOI:10.1038/s41541-022-00429-5.
[27] Hahn BL,Padmore LJ,Ristow LC,et al. Live attenuated Borrelia burgdorferi targeted mutants in an infectious strain background protect mice from challenge infection[J]. Clin Vaccine Immunol,2016,23(8):725-731. DOI:10.1128/CVI.00302-16.
[28] Ding W,Huang XL,Yang XH,et al. Structural identification of a key protective B-cell epitope in Lyme disease antigen OspA[J]. J Mol Biol,2000,302(5):1153-1164. DOI:10.1006/jmbi.2000. 4119.
[29] Vogt NA,Sargeant JM,Mackinnon MC,et al. Efficacy of Borrelia burgdorferi vaccine in dogs in North America:A systematic review and meta-analysis[J]. J Vet Intern Med,2019,33(1):23-36. DOI:10.1111/jvim.15344.
[30] Guibinga GH,Sahay B,Brown H,et al. Protection against Borreliella burgdorferi infection mediated by a synthetically engineered DNA vaccine[J]. Hum Vaccin Immunother,2020,16(9):2114-2122. DOI:10.1080/21645515.2020.1789408.
[31] 贺琪楠,尹家祥.鼠型斑疹伤寒流行现状及其影响因素[J].中国热带医学,2019,19(8):790-793. DOI:10.13604/j.cnki.46-1064/r.2019.08.19. He QN,Yin JX. Epidemic situation and influential factors of murine typhus[J]. China Trop Med,2019,19(8):790-793. DOI:10.13604/j.cnki.46-1064/r.2019.08.19.(in Chinese)
[32] Osterloh A. The neglected challenge:Vaccination against rickettsiae[J]. PLoS Negl Trop Dis,2020,14(10):e0008704. DOI:10.1371/journal.pntd.0008704.
[33] Walker DH. The realities of biodefense vaccines against Rickettsia[J]. Vaccine,2009,27 Suppl 4:D52-D55. DOI:10. 1016/j.vaccine.2009.07.045.
[34] Choi S,Jeong HJ,Hwang KJ,et al. A recombinant 47-kDa outer membrane protein induces an immune response against Orientia tsutsugamushi strain Boryong[J]. Am J Trop Med Hyg,2017,97(1):30-37. DOI:10.4269/ajtmh.15-0771.
[35] Kim HI,Ha NY,Kim G,et al. Immunization with a recombinant antigen composed of conserved blocks from TSA56 provides broad genotype protection against scrub typhus[J]. Emerg Microbes Infect,2019,8(1):946-958. DOI:10.1080/22221751. 2019.1632676.
[36] Cho H,Lee WH,Kim YK,et al. Extracellular vesicle-associated antigens as a new vaccine platform against scrub typhus[J]. Biochem Biophys Res Commun,2020,523(3):602-607. DOI:10.1016/j.bbrc.2020.01.014.
[37] Song SW,Kim KT,Ku YM,et al. Clinical role of interstitial pneumonia in patients with scrub typhus:A possible marker of disease severity[J]. J Korean Med Sci,2004,19(5):668-673. DOI:10.3346/jkms.2004.19.5.668.
[38] Park SM,Gu MJ,Ju YJ,et al. Intranasal vaccination with outer-membrane protein of Orientia tsutsugamushi induces protective immunity against Scrub Typhus[J]. Immune Netw,2021,21(2):e14. DOI:10.4110/in.2021.21.e14.
[39] Liu RR,Ma HW,Shu JY,et al. Vaccines and therapeutics against Hantaviruses[J]. Front Microbiol,2020,10:2989. DOI:10.3389/fmicb.2019.02989.
[40] Mir S. Hantavirus induced kidney disease[J]. Front Med (Lausanne),2022,8:795340. DOI:10.3389/fmed.2021.795340.