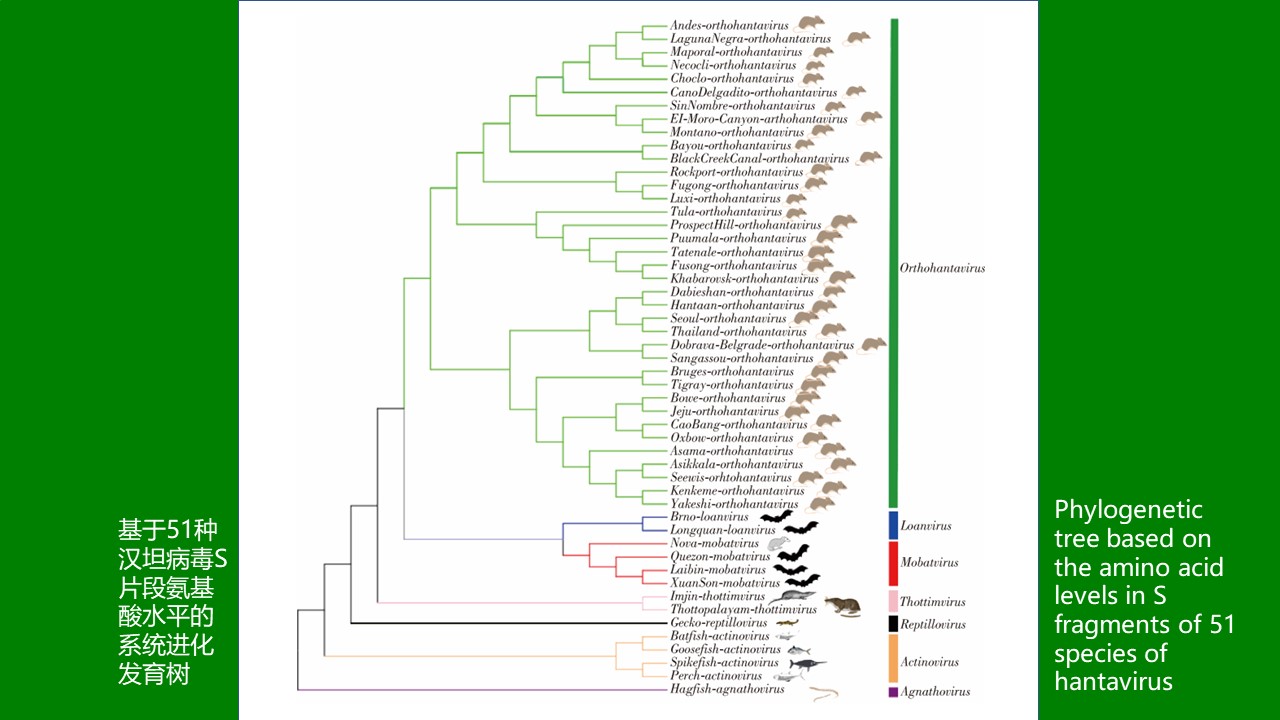
Hantaviruses are important pathogenes of natural focal diseases that causes hemorrhagic fever with renal syndrome and Hantavirus pulmonary syndrome. According to the latest classification of the International Committee on Taxonomy of Viruses, hantaviruses can be divided into 53 species, 7 genera, and 4 subfamilies. Hantaviruses are widely found in Rodentia, Chiroptera, and Insectivora, and later also found in reptile, Actinopterygii, and Agnatha. There are many species of bats, which are the second largest group of mammals in the world after rodents. At present, 1 446 species have been reported, accounting for about 22% of global mammals. In addition, bats have strong flight ability and are widely distributed in all continents except Antarctica. As the host animal of viruses, bats bear a variety of viruses, and many emerging infectious pathogens such as Marburg virus, Hendra virus, and Nipah virus have been confirmed to come from bats. Bats have also been associated to Ebola virus, severe acute respiratory syndrome coronavirus, Middle East respiratory syndrome coronavirus, and severe acute respiratory syndrome coronavirus 2. In recent decades, more and more bat-borne hantaviruses have been discovered. Bats and bat-borne hantaviruses have an important research value. Therefore, this paper reviews the latest classification of Hantavirus and bat-borne hantaviruses.
[1] French GR, Foulke RS, Brand OA, et al. Korean hemorrhagic fever:Propagation of the etiologic agent in a cell line of human origin[J]. Science, 1981, 211(4486):1046-1048. DOI:10.1126/science.6110243.
[2] Zhang YZ. Discovery of hantaviruses in bats and insectivores and the evolution of the genus Hantavirus[J]. Virus Res, 2014, 187:15-21. DOI:10.1016/j.virusres.2013.12.035.
[3] ASM Mammal Diversity Database[EB/OL]. (2021-10-09)[2021-12-01]. http://www.mammaldiversity.org/.
[4] Baker RJ, Longmire JL, Maltbie M, et al. DNA synapomorphies for a variety of taxonomic levels from a cosmid library from the New World BAT Macrotus waterhousii[J]. Syst Biol, 1997, 46(4):579-589. DOI:10.1093/sysbio/46.4.579.
[5] Hutcheon JM, Kirsch JAW, Pettigrew JD. Base-compositional biases and the bat problem. III. The question of microchiropteran monophyly[J]. Philos Trans R Soc Lond B Biol Sci, 1998, 353(1368):607-617. DOI:10.1098/rstb.1998.0229.
[6] Teeling EC, Scally M, Kao DJ, et al. Molecular evidence regarding the origin of echolocation and flight in bats[J]. Nature, 2000, 403(6766):188-192. DOI:10.1038/35003188.
[7] Teeling EC, Madsen O, Murphy WJ, et al. Nuclear gene sequences confirm an ancient link between New Zealand's short-tailed bat and South American noctilionoid bats[J]. Mol Phylogenet Evol, 2003, 28(2):308-319. DOI:10.1016/s1055-7903(03)00117-9.
[8] Calisher CH, Childs JE, Field HE, et al. Bats:Important reservoir hosts of emerging viruses[J]. Clin Microbiol Rev, 2006, 19(3):531-545. DOI:10.1128/CMR.00017-06.
[9] Schönrich G, Rang A, Lütteke N, et al. Hantavirus-induced immunity in rodent reservoirs and humans[J]. Immunol Rev, 2008, 225(1):163-189. DOI:10.1111/j.1600-065X.2008.00694.x.
[10] Baker ML, Schountz T, Wang LF. Antiviral immune responses of bats:A review[J]. Zoonoses Public Health, 2013, 60(1):104-116. DOI:10.1111/j.1863-2378.2012.01528.x.
[11] Moratelli R, Calisher CH. Bats and zoonotic viruses:Can we confidently link bats with emerging deadly viruses?[J]. Mem Inst Oswaldo Cruz, 2015, 110(1):1-22. DOI:10.1590/0074-02760150048.
[12] Wu ZQ, Yang L, Ren XW, et al. Deciphering the bat virome catalog to better understand the ecological diversity of bat viruses and the bat origin of emerging infectious diseases[J]. ISME J, 2016, 10(3):609-620. DOI:10.1038/ismej.2015.138.
[13] Letko M, Seifert SN, Olival KJ, et al. Bat-borne virus diversity, spillover and emergence[J]. Nat Rev Microbiol, 2020, 18(8):461-471. DOI:10.1038/s41579-020-0394-z.
[14] Olival KJ, Hayman DTS. Filoviruses in bats:Current knowledge and future directions[J]. Viruses, 2014, 6(4):1759-1788. DOI:10.3390/v6041759.
[15] Memish ZA, Mishra N, Olival KJ, et al. Middle east respiratory syndrome coronavirus in bats, Saudi Arabia[J]. Emerg Infect Dis, 2013, 19(11):1819-1823. DOI:10.3201/eid1911.131172.
[16] Leroy EM, Kumulungui B, Pourrut X, et al. Fruit bats as reservoirs of Ebola virus[J]. Nature, 2005, 438(7068):575-576. DOI:10.1038/438575a.
[17] Amman BR, Albariño CG, Bird BH, et al. A recently discovered pathogenic paramyxovirus, sosuga virus, is present in Rousettus aegyptiacus fruit bats at multiple locations in Uganda[J]. J Wildl Dis, 2015, 51(3):774-779. DOI:10.7589/2015-02-044.
[18] Arai S, Yanagihara R. Genetic diversity and geographic distribution of bat-borne hantaviruses[J]. Curr Issues Mol Biol, 2020, 39:1-28. DOI:10.21775/cimb.039.001.
[19] Holmes EC, Zhang YZ. The evolution and emergence of hantaviruses[J]. Curr Opin Virol, 2015, 10:27-33. DOI:10. 1016/j.coviro.2014.12.007.
[20] Brook CE, Dobson AP. Bats as ‘special’ reservoirs for emerging zoonotic pathogens[J]. Trends Microbiol, 2015, 23(3):172-180. DOI:10.1016/j.tim.2014.12.004.
[21] Tadin A, Turk N, Korva M, et al. Multiple co-infections of rodents with hantaviruses, Leptospira, and Babesia in Croatia[J]. Vector Borne Zoonotic Dis, 2012, 12(5):388-392. DOI:10.1089/vbz.2011.0632.
[22] Vapalahti O, Mustonen J, Lundkvist A, et al. Hantavirus infections in Europe[J]. Lancet Infect Dis, 2003, 3(10):653-661. DOI:10.1016/s1473-3099(03)00774-6.
[23] Heyman P, Vaheri A, Lundkvist A, et al. Hantavirus infections in Europe:From virus carriers to a major public-health problem[J]. Expert Rev Anti-Infect Ther, 2009, 7(2):205-217. DOI:10. 1586/14787210.7.2.205.
[24] MacNeil A, Nichol ST, Spiropoulou CF. Hantavirus pulmonary syndrome[J]. Virus Res, 2011, 162(1/2):138-147. DOI:10. 1016/j.virusres.2011.09.017.
[25] Guo WP, Lin XD, Wang W, et al. Phylogeny and origins of hantaviruses harbored by bats, insectivores, and rodents[J]. PLoS Pathog, 2013, 9(2):e1003159. DOI:10.1371/journal.ppat. 1003159.
[26] Straková P, Dufkova L, Širmarová J, et al. Novel hantavirus identified in European bat species Nyctalus noctula[J]. Infect Genet Evol, 2017, 48:127-130. DOI:10.1016/j.meegid.2016. 12.025.
[27] Xu L, Wu JM, He B, et al. Novel hantavirus identified in black-bearded tomb bats, China[J]. Infect Genet Evol, 2015, 31:158-160. DOI:10.1016/j.meegid.2015.01.018.
[28] Arai S, Kikuchi F, Bawm S, et al. Molecular phylogeny of mobatviruses (Hantaviridae) in Myanmar and Vietnam[J]. Viruses, 2019, 11(3):228. DOI:10.3390/v11030228.
[29] Arai S, Nguyen ST, Boldgiv B, et al. Novel bat-borne hantavirus, Vietnam[J]. Emerg Infect Dis, 2013, 19(7):1159-1161. DOI:10.3201/eid1907.121549.
[30] Arai S, Taniguchi S, Aoki K, et al. Molecular phylogeny of a genetically divergent hantavirus harbored by the Geoffroy's rousette (Rousettus amplexicaudatus), a frugivorous bat species in the Philippines[J]. Infect Genet Evol, 2016, 45:26-32. DOI:10.1016/j.meegid.2016.08.008.
[31] de Araujo J, Thomazelli LM, Henriques DA, et al. Detection of hantavirus in bats from remaining rain forest in São Paulo, Brazil[J]. BMC Res Notes, 2012, 5:690. DOI:10.1186/1756-0500-5-690.
[32] Sabino-Santos G Jr, Maia FGM, Vieira TM, et al. Evidence of hantavirus infection among bats in Brazil[J]. Am J Trop Med Hyg, 2015, 93(2):404-406. DOI:10.4269/ajtmh.15-0032.
[33] Sabino-Santos G Jr, Maia FGM, Martins RB, et al. Natural infection of Neotropical bats with hantavirus in Brazil[J]. Sci Rep, 2018, 8(1):9018. DOI:10.1038/s41598-018-27442-w.
[34] Yanagihara R, Gu SH, Arai S, et al. Hantaviruses:Rediscovery and new beginnings[J]. Virus Res, 2014, 187:6-14. DOI:10. 1016/j.virusres.2013.12.038.
[35] Bennett SN, Gu SH, Kang HJ, et al. Reconstructing the evolutionary origins and phylogeography of hantaviruses[J]. Trends Microbiol, 2014, 22(8):473-482. DOI:10.1016/j.tim. 2014.04.008.
[36] Witkowski PT, Drexler JF, Kallies R, et al. Phylogenetic analysis of a newfound bat-borne hantavirus supports a laurasiatherian host association for ancestral mammalian hantaviruses[J]. Infect Genet Evol, 2016, 41:113-119. DOI:10.1016/j.meegid.2016. 03.036.



