

廖承红,E-mail:liaochh@hainanu.edu.cn
LIAO Cheng-hong, E-mail: liaochh@hainanu.edu.cn
韩谦,E-mail:qianhan@hainanu.edu.cn
HAN Qian, E-mail: qianhan@hainanu.edu.cn
收稿日期: 2023-02-06
网络出版日期: 2023-08-17
基金资助
国家自然科学基金(U22A20363);海南省重大科技计划项目(ZDKJ2021035);海南省重点研发项目(ZDYF2023XDNY061)
A metagenomic analysis of the composition of symbiotic microbial communities in Culicoides on different livestock farms in Hainan Province
Received date: 2023-02-06
Online published: 2023-08-17
Supported by
National Natural Science Foundation of China(U22A20363);The Major Science and Technology Plan of Hainan Province(ZDKJ2021035);Hainan Provincial Key Research Program(ZDYF2023XDNY061)
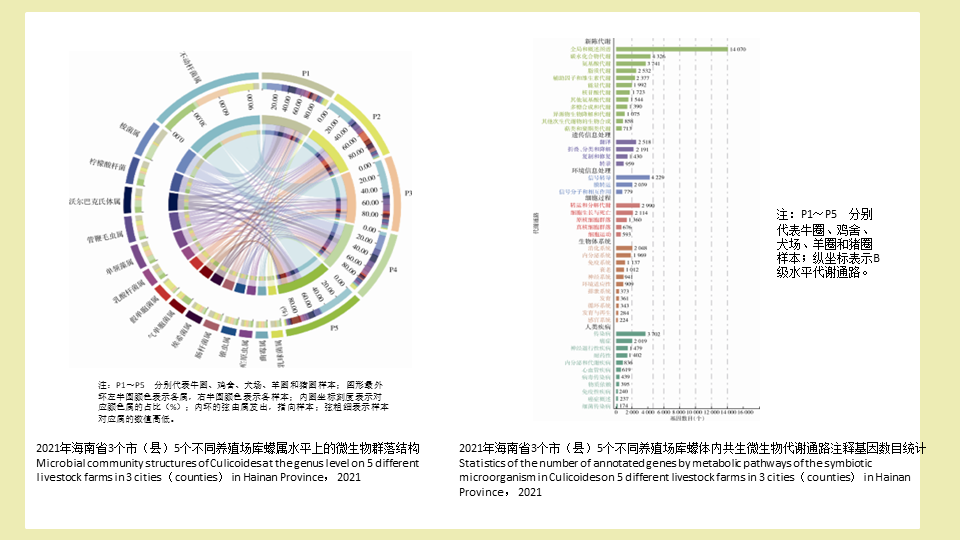
目的: 探究海南省不同养殖场库蠓共生微生物群落多样性和差异性。方法: 2021年8-9月,在海南省文昌市、定安和乐东县5个不同的养殖场(牛圈、鸡舍、犬场、羊圈和猪圈),用紫外灯诱法采集库蠓,分别使用十六烷基三甲基溴化铵法提取总DNA后,通过宏基因组学测序技术分析其微生物群落结构特征及基因功能分布。结果: 不同养殖场库蠓优势种不同,牛圈、羊圈和猪圈主要是尖喙库蠓,占比分别为92.91%,82.22%和95.85%;鸡舍和犬场均以荒川库蠓为主要优势种,占比分别为66.36%和98.22%。不同养殖场库蠓共生微生物在门水平上,均以变形菌门(33.78%)、子囊菌门(9.09%)和厚壁菌门(8.88%)为主要菌门;在属水平上,均以不动杆菌属(22.48%)和梭菌属(7.25%)为优势菌属。属水平微生物多样性分析发现5个养殖场共有的属最多,有3 357属。微生物基因差异分析显示5个不同的养殖场共有基因数目较少,为1 065个。此外,功能预测结果也显示,29.10%注释基因与新陈代谢相关,如碳水化合物代谢、氨基酸代谢和脂质代谢等。基因数目差异分析结果表明,库蠓种类组成越相似,样品共有基因数目越多。结论: 微生物群落分析发现,海南省不同养殖场库蠓共生微生物的核心菌群相对稳定,库蠓优势种相同的养殖场微生物群落相似度更高。该研究丰富了我国库蠓的微生物群落数据库,对今后库蠓的生物防控有重要意义。
樊洁丽, 刘焱晖, 殷雅楠, 赵建国, 孙定炜, 廖承红, 韩谦 . 基于宏基因组学分析海南省不同养殖场库蠓共生微生物群落组成[J]. 中国媒介生物学及控制杂志, 2023 , 34(4) : 472 -479 . DOI: 10.11853/j.issn.1003.8280.2023.04.006
Objective: To explore the diversity and variability of symbiotic microbial communities in Culicoides on different livestock farms in Hainan Province, China. Methods: From August to September 2021, the ultraviolet lamp-trap method was used to collect Culicoides on five different livestock farms (cattle farms, chicken houses, dog houses, sheep pens, and pigsties) in Wenchang City, Ding'an County, and Ledong County in Hainan Province. After the total DNA was extracted by hexadecyltrimethylammonium bromide method, the structural characteristics and functional distribution of genes in the microbial communities were analyzed using metagenomic sequencing. Results: The dominant species of Culicoides on different livestock farms varied. The dominant species on cattle farms, sheep pens, and pigsties was C. oxystoma, accounting for 92.91%, 82.22%, and 95.85%, respectively; the dominant species in chicken houses and dog houses were C. arakawae, accounting for 66.36% and 98.22%, respectively. On different livestock farms, Proteobacteria (33.78%), Ascomycota (9.09%), and Firmicutes (8.88%) were the dominant phyla of the symbiotic microbial communities in Culicoides, and Acinetobacter (22.48%) and Clostridium (7.25%) were the dominant genera. Microbial diversity analysis at the genus level found that the five livestock farms shared the most microorganisms at the genus level, with 3 357 genera in common. Microbial gene differential analysis revealed that the five livestock farms shared a relatively small number of common genes (1 065). Moreover, the functional prediction results also showed that 29.10% of the annotated genes were related to metabolism, such as carbohydrate metabolism, amino acid metabolism, and lipid metabolism. The analysis of the differences in gene number showed that increased similarity in the species composition of Culicoides was associated with a greater number of genes shared by the samples. Conclusions: Analysis of the microbial communities found that the core symbiotic microflora communities in Culicoides is relatively stable, and livestock farms with the same dominant species of Culicoides have more similarities in microbial communities in Culicoides. This study enriches the microbial community database of Culicoides in China, which is of great significance for the biological prevention and control of Culicoides in the future.

Key words: Culicoides; Symbiotic microorganism; Metagenomics
| 1 | Borkent A, Dominiak P. Catalog of the biting midges of the world (Diptera: Ceratopogonidae)[J]. Zootaxa, 2020, 4787 (1):1-377. |
| 2 | 霍坚, 高永利, 郑龙, 等. 土拉弗朗西斯菌病的特征与防治[J]. 中华卫生杀虫药械, 2021, 27 (2):182-186. |
| 2 | Huo J, Gao YL, Zheng L, et al. Research progress of characteristics, prevention and treatment on tularemia[J]. Chin J Hyg Insect Equip, 2021, 27 (2):182-186. |
| 3 | Yu CY, Wang JS, Yeh CC. Culicoides arakawae (Diptera: Ceratopogonidae) population succession in relation to leucocytozoonosis prevalence on a chicken farm in Taiwan[J]. Vet Parasitol, 2000, 93 (2):113-120. |
| 4 | Duan YL, Li L, Bellis G, et al. Detection of Bluetongue virus in Culicoides spp. in southern Yunnan province, China[J]. Parasit Vectors, 2021, 14 (1):68. |
| 5 | Ségard A, Gardès L, Jacquier E, et al. Schmallenberg virus in Culicoides Latreille (Diptera: Ceratopogonidae) populations in France during 2011-2012 outbreak[J]. Transbound Emerg Dis, 2018, 65 (1):e94-e103. |
| 6 | Mcgregor BL, Shults PT, Mcdermott EG. A review of the vector status of North American Culicoides (Diptera: Ceratopogonidae) for Bluetongue virus, Epizootic hemorrhagic disease virus, and other arboviruses of concern[J]. Curr Trop Med Rep, 2022, 9 (4):130-139. |
| 7 | 刘国平, 孙定炜, 范娜, 等. 海南省5市县禽畜厩舍吸血蠓调查[J]. 中国热带医学, 2020, 20 (5):413-416, 428. |
| 7 | Liu GP, Sun DW, Fan N, et al. Hematophagous midges at the hencoop and livestock sheds in five counties of Hainan[J]. China Trop Med, 2020, 20 (5):413-416, 428. |
| 8 | 叶雅芳, 刘德星, 李婷婷, 等. 广东省中山市库蠓形态与分子鉴定[J]. 中国人兽共患病学报, 2019, 35 (11):1021-1028. |
| 8 | Ye YF, Liu DX, Li TT, et al. Morphological and molecular identification of Culicoides collected in Zhongshan, Guangdong, China[J]. Chin J Zoonoses, 2019, 35 (11):1021-1028. |
| 9 | 刘帅, 加帕尔·哈斯木, 薛新梅, 等. 新疆库蠓源性盖塔病毒的分离与鉴定[J]. 畜牧兽医学报, 2017, 48 (10):1998-2004. |
| 9 | Liu S, Jiapaer HSM, Xue XM, et al. Isolation and identification of Getah virus from Culicoides in Xinjiang[J]. Acta Veterin Zootechn Sin, 2017, 48 (10):1998-2004. |
| 10 | Duan YL, Yang ZX, Bellis G, et al. Isolation of Tibet orbivirus from Culicoides jacobsoni (Diptera, Ceratopogonidae) in China[J]. Parasit Vectors, 2021, 14 (1):432. |
| 11 | Storelli G, Defaye A, Erkosar B, et al. Lactobacillus plantarum promotes Drosophila systemic growth by modulating hormonal signals through TOR-dependent nutrient sensing[J]. Cell Metab, 2011, 14 (3):403-414. |
| 12 | Cirstea MS, Yu AC, Golz E, et al. Microbiota composition and metabolism are associated with gut function in Parkinson's disease[J]. Mov Disord, 2020, 35 (7):1208-1217. |
| 13 | 周洪英, 孙波, 吴洪丽, 等. 昆虫肠道微生物功能及家蚕肠道微生物研究进展[J]. 北方蚕业, 2015, 36 (4):1-4, 33. |
| 13 | Zhou HY, Sun B, Wu HL, et al. Research progress on function of insects gut microbiota and the microbial of Bombyx mori[J]. North Sericult, 2015, 36 (4):1-4, 33. |
| 14 | 索鹏辉, 符修昊, 安丽萍, 等. 海南省儋州市旱季致倦库蚊肠道细菌多样性研究[J]. 中国热带医学, 2022, 22 (1):20-25. |
| 14 | Suo PH, Fu XH, An LP, et al. The diversity of midgut bacteria of Culex quinquefasciatus in the dry season in Danzhou, Hainan[J]. China Trop Med, 2022, 22 (1):20-25. |
| 15 | 夏晓峰. 小菜蛾中肠微生物多样性及其功能研究[D]. 福州: 福建农林大学, 2014. |
| 15 | Xia XF. Organizational diversity and functional characterization of microbiota in the midgut of diamondback moth, Plutella xylostella (L.)[D]. Fuzhou: Fujian Agriculture and Forestry University, 2014. (in Chinese) |
| 16 | Pasti MB, Belli ML. Cellulolytic activity of Actinomycetes isolated from termites (Termitidae) gut[J]. FEMS Microbiol Lett, 1985, 26 (1):107-112. |
| 17 | Waite DW, Taylor MW. Characterizing the avian gut microbiota: Membership, driving influences, and potential function[J]. Front Microbiol, 2014, 5, 223. |
| 18 | Klement RJ, Pazienza V. Impact of different types of diet on gut microbiota profiles and cancer prevention and treatment[J]. Medicina (Kaunas), 2019, 55 (4):84. |
| 19 | Boyle DP, Zembower TR. Epidemiology and management of emerging drug-resistant gram-negative bacteria: Extended-spectrum β-lactamases and beyond[J]. Urol Clin North Am, 2015, 42 (4):493-505. |
| 20 | Boissière A, Tchioffo MT, Bachar D, et al. Midgut microbiota of the malaria mosquito vector Anopheles gambiae and interactions with Plasmodium falciparum infection[J]. PLoS Pathog, 2012, 8 (5):e1002742. |
| 21 | Werren JH. Biology of wolbachia[J]. Ann Rev Entomol, 1997, 42, 587-609. |
| 22 | Jaenike J. Spontaneous emergence of a new Wolbachia phenotype[J]. Evolution, 2007, 61 (9):2244-2252. |
| 23 | Sunantaraporn S, Thepparat A, Phumee A, et al. Culicoides Latreille (Diptera: Ceratopogonidae) as potential vectors for Leishmania martiniquensis and Trypanosoma sp. in northern Thailand[J]. PLoS Negl Trop Dis, 2021, 15 (12):e0010014. |
| 24 | Kohl KD. Diversity and function of the avian gut microbiota[J]. J Comp Physiol B, 2012, 182 (5):591-602. |
/
| 〈 |
|
〉 |