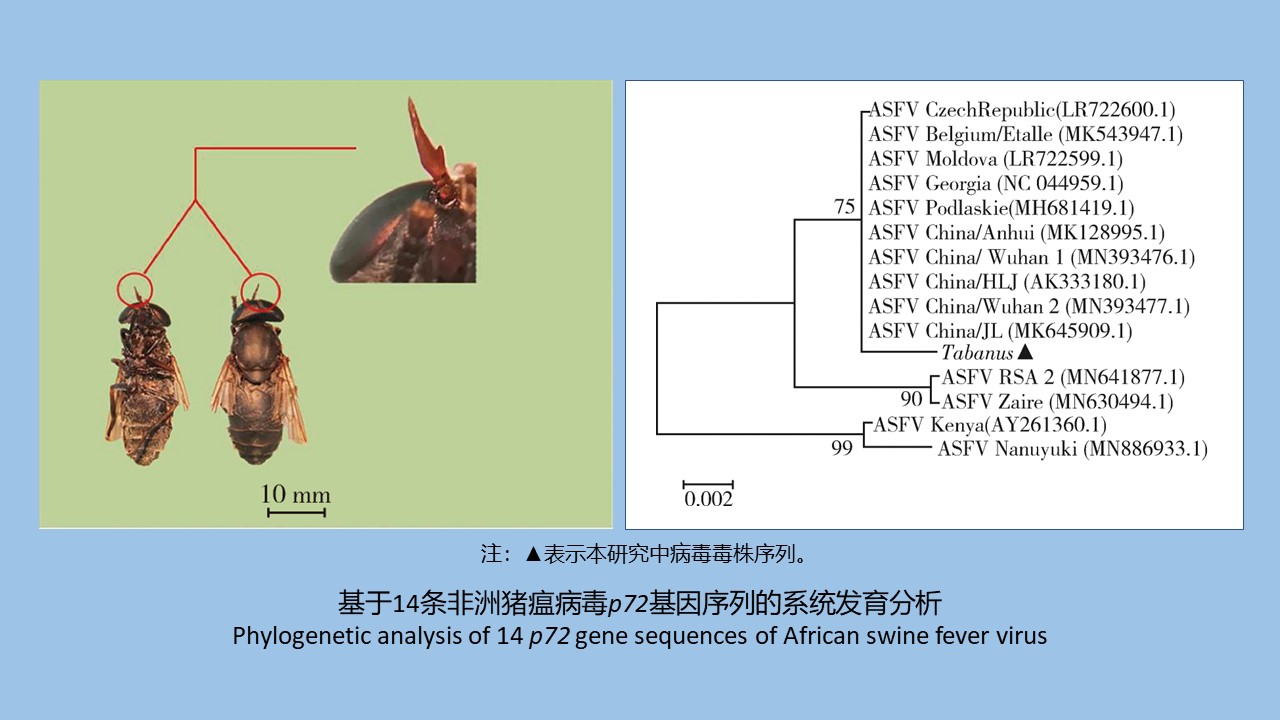
目的 了解安徽省金寨县大别山地区虻科食血昆虫在非洲猪瘟病毒(ASFV)传播中的作用,为该地区非洲猪瘟防控提供依据。方法 2019年9月在安徽省金寨县大别山地区采用昆虫诱捕方法收集野生虻。对采集到的野生虻用体视显微镜观察,基于形态分类学特征对其进行初步种属鉴定。对采集到的野生虻进行分体研磨,组织核酸提取试剂盒和病毒核酸提取试剂盒用于提取DNA。野生虻组织基因组采用线粒体细胞色素C氧化酶亚基Ⅰ(COⅠ)基因引物扩增并进行测序及序列分析以鉴定其种属。基于世界动物卫生组织公布的ASFV p72基因引物对病毒核酸进行PCR扩增、测序及序列分析,检测分析野生虻中ASFV是否存在。结果 在3处不同地点共采集24只野生虻,基于形态分类学观察以及COⅠ基因常规扩增后测序,野生虻鉴定为Tabanus hypomacros。ASFV p72基因引物对部分病毒核酸提取样本扩增阳性,ASFV的总阳性检出率达45.83%。ASFV p72基因阳性扩增产物测序获得278 nt序列。系统进化分析结果显示,野生虻中病毒与ASFV中国流行株处于同一进化分支内。结论 在安徽省金寨县大别山地区采集的野生虻中检测到ASFV且与中国流行株高度同源,野生虻T.hypomacros可作为ASFV潜在的机械传播媒介。
Objective To investigate the role of blood-eating insects in the family Tabanidae in the spread of African swine fever virus (ASFV) in the Dabie Mountain region of Jinzhai county, Anhui province, China, and to provide evidence for the prevention and control of Africian swine fever in the region. Methods The insect trap method was used to collect wild horsefly in the Dabie Mountain region of Jinzhai county, Anhui province in September 2019. The collected horseflies were observed with a stereo microscope to preliminarily identify their species based on their morphological characteristics. The collected horseflies were separately ground and DNA was extracted using a tissue nucleic acid extraction kit and a virus nucleic acid extraction kit. Cytochrome c oxidase subunit Ⅰ (COⅠ) gene amplification and sequencing were performed on the tissue genome to identify the species of the horseflies. With the ASFV p72 gene primers published by World Organisation for Animal Health, the ASFV nucleic acid was amplified by PCR, followed by sequencing to detect the DNA of ASFV in the horsefly samples. Results A total of 24 horseflies were collected from three different locations. Through morphological observations and COⅠgene amplification and sequencing, the horseflies were identified as Tabanus hypomacros. Amplification with the ASFV p72 gene primers was positive for part of viral nucleic acid extraction samples, with an overall positive detection rate of ASFV of 45.83%. The product of the positive amplification with ASFV p72 primers was sequenced to be a 278 nt sequence. The phylogenetic analysis showed that the ASFV in the horsefly specimens and the epidemic ASFV strains in China were in the same evolutionary branch. Conclusion ASFV is found in wild horseflies in the Dabie Mountain region of Jinzhai county, Anhui province, and is highly homologous to the epidemic ASFV strains in China, indicating that T. hypomacros can be a potential mechanical transmission vector of ASFV.
[1] 彭志锋, 卢建洲, 蒋增海, 等. 非洲猪瘟病毒疫苗的研究进展[J]. 中国兽医学报, 2020, 40(4):859-863. DOI:10.16303/j.cnki.1005-4545.2020.04.33. Peng ZF, Lu JZ, Jiang ZH, et al. Progress and perspectives for development of African swine fever virus vaccines[J]. Chin J Vet Sci, 2020, 40(4):859-863. DOI:10.16303/j.cnki.1005-4545.2020.04.33.(in Chinese)
[2] Sánchez-Vizcaíno JM, Mur L, Gomez-Villamandos JC, et al. An update on the epidemiology and pathology of African swine fever[J]. J Comp Pathol, 2015, 152(1):9-21. DOI:10.1016/j.jcpa.2014.09.003.
[3] Davies K, Goatley LC, Guinat C, et al. Survival of African swine fever virus in excretions from pigs experimentally infected with the Georgia 2007/1 isolate[J]. Transbound Emerg Dis, 2017, 64(2):425-431. DOI:10.1111/tbed.12381.
[4] Fasina FO, Shamaki D, Makinde AA, et al. Surveillance for African swine fever in Nigeria, 2006-2009[J]. Transbound Emerg Dis, 2010, 57(4):244-253. DOI:10.1111/j.1865-1682.2010.01142.x.
[5] Zhou XT, Li N, Luo YZ, et al. Emergence of African swine fever in China, 2018[J]. Transbound Emerg Dis, 2018, 65(6):1482-1484. DOI:10.1111/tbed.12989.
[6] Sánchez-Cordón PJ, Montoya M, Reis AL, et al. African swine fever:A re-emerging viral disease threatening the global pig industry[J]. Vet J, 2018, 233:41-48. DOI:10.1016/j.tvjl. 2017.12.025.
[7] Mur L, Martínez-López B, Sánchez-Vizcaíno JM. Risk of African swine fever introduction into the European Union through transport-associated routes:Returning trucks and waste from international ships and planes[J]. BMC Vet Res, 2012, 8:149. DOI:10.1186/1746-6148-8-149.
[8] Golnar AJ, Martin E, Wormington JD, et al. Reviewing the potential vectors and hosts of African swine fever virus transmission in the United States[J]. Vector Borne Zoonotic Dis, 2019, 19(7):512-524. DOI:10.1089/vbz.2018.2387.
[9] Olesen AS, Lohse L, Boklund A, et al. Transmission of African swine fever virus from infected pigs by direct contact and aerosol routes[J]. Vet Microbiol, 2017, 211:92-102. DOI:10.1016/j.vetmic.2017.10.004.
[10] Gallardo C, Soler A, Nieto R, et al. Experimental infection of domestic pigs with African swine fever virus Lithuania 2014 genotype II field isolate[J]. Transbound Emerg Dis, 2017, 64(1):300-304. DOI:10.1111/tbed.12346.
[11] Plowright W, Parker J, Peirce MA. African swine fever virus in ticks (Ornithodoros moubata, Murray) collected from animal burrows in Tanzania[J]. Nature, 1969, 221(5185):1071-1073. DOI:10.1038/2211071a0.
[12] Chen Z, Xu XF, Wang YF, et al. DNA segments of African swine fever virus detected for the first time in hard ticks from sheep and bovines[J]. Syst Appl Acarol, 2019, 24(1):180. DOI:10.11158/saa.24.1.13.
[13] Olesen AS, Hansen MF, Rasmussen TB, et al. Survival and localization of African swine fever virus in stable flies (Stomoxys calcitrans) after feeding on viremic blood using a membrane feeder[J]. Vet Microbiol, 2018, 222:25-29. DOI:10.1016/j.vetmic.2018.06.010.
[14] De Liberato C, Magliano A, Autorino GL, et al. Seasonal succession of tabanid species in equine infectious anaemia endemic areas of Italy[J]. Med Vet Entomol, 2019, 33(3):431-436. DOI:10.1111/mve.12360.
[15] Al-Talafha H, Yaakop SB, Ghani IBA. Taxonomic notes and new records of the genus Tabanus Linnaeus 1758(Diptera:Tabanidae) from Malaysia[J]. AIP Conf Proc, 2016, 1784(1):060014. DOI:10.1063/1.4966852.
[16] Hebert PDN, Penton EH, Burns JM, et al. Ten species in one:DNA barcoding reveals cryptic species in the neotropical skipper butterfly Astraptes fulgerator[J]. Proc Natl Acad Sci USA, 2004, 101(41):14812-14817. DOI:10.1073/pnas.0406166101.
[17] 中华人民共和国国家质量监督检验检疫总局. SN/T 1559-2010非洲猪瘟检疫技术规范[S]. 北京:中国标准出版社, 2010. General Administration of Quality Supervision, Inspection and Quarantine of the People's Republic of China. SN/T 1559-2010 Protocol of quarantine for African swine fever[S]. Beijing:Standards Press of China, 2010. (in Chinese)
[18] Baldacchino F, Gardès L, De Stordeur E, et al. Blood-feeding patterns of horse flies in the French Pyrenees[J]. Vet Parasitol, 2014, 199(3/4):283-288. DOI:10.1016/j.vetpar.2013.10.009.
[19] Baldacchino F, Desquesnes M, Mihok S, et al. Tabanids:Neglected subjects of research, but important vectors of disease agents![J]. Infect Genet Evol, 2014, 28:596-615. DOI:10.1016/j.meegid.2014.03.029.
[20] Mellor PS, Kitching RP, Wilkinson PJ. Mechanical transmission of capripox virus and African swine fever virus by Stomoxys calcitrans[J]. Res Vet Sci, 1987, 43(1):109-112. DOI:10.1016/S0034-5288(18)30753-7.
[21] Salem A, Franc M, Jacquiet P, et al. Feeding and breeding aspects of Stomoxys calcitrans (Diptera:Muscidae) under laboratory conditions[J]. Parasite, 2012, 19(4):309-317. DOI:10.1051/parasite/2012194309.
[22] De Oliveira RP, Hutet E, Lancelot R, et al. Differential vector competence of Ornithodoros soft ticks for African swine fever virus:What if it involves more than just crossing organic barriers in ticks?[J]. Parasit Vectors, 2020, 13(1):618. DOI:10.1186/S13071-020-04497-1.
[23] Cywinska A, Hannan MA, Kevan PG, et al. Evaluation of DNA barcoding and identification of new haplomorphs in Canadian deerflies and horseflies[J]. Med Vet Entomol, 2010, 24(4):382-410. DOI:10.1111/j.1365-2915.2010.00896.x.



