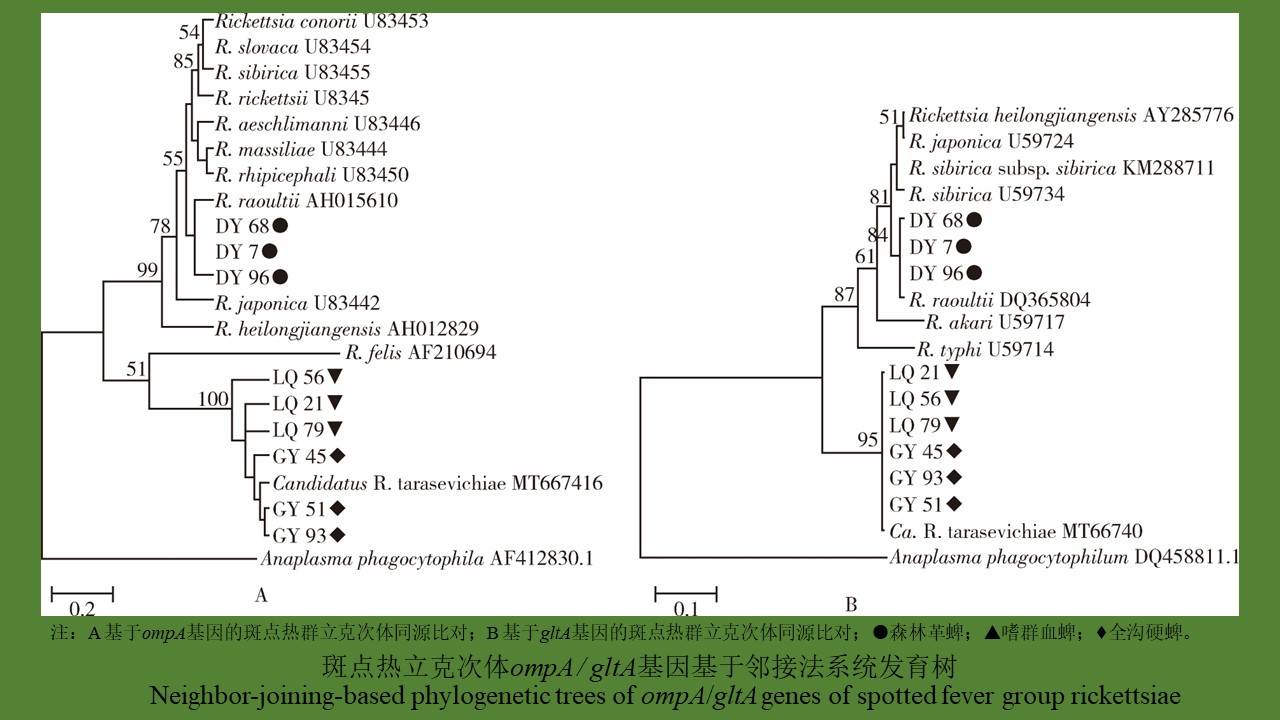
[1] Merhej V, Angelakis E, Socolovschi C, et al. Genotyping, evolution and epidemiological findings of Rickettsia species[J]. Infect Genet Evol, 2014, 25:122-137. DOI:10.1016/j.meegid. 2014.03.014.
[2] Vanegas A, Keller C, Krüger A, et al. Molecular detection of spotted fever group rickettsiae in ticks from Cameroon[J]. Ticks Tick-Borne Dis, 2018, 9(4):1049-1056. DOI:10.1016/j.ttbdis. 2018.03.022.
[3] Fang LQ, Liu K, Li XL, et al. Emerging tick-borne infections in mainland China:An increasing public health threat[J]. Lancet Infect Dis, 2015, 15(12):1467-1479. DOI:10.1016/S1473-3099(15)00177-2.
[4] Parola P, Paddock CD, Socolovschi C, et al. Update on tick-borne rickettsioses around the world:A geographic approach[J]. Clin Microbiol Rev, 2014, 27(1):657-702. DOI:10.1128/CMR. 00104-13.
[5] Wei QQ, Guo LP, Wang AD, et al. The first detection of Rickettsia aeschlimannii and R. massiliae in Rhipicephalus turanicus ticks, in northwest China[J]. Parasit Vectors, 2015, 8(1):631. DOI:10.1186/s13071-015-1242-2.
[6] Yang JF, Tian ZC, Liu ZJ, et al. Novel spotted fever group rickettsiae in Haemaphysalis qinghaiensis ticks from Gansu, Northwest China[J]. Parasit Vectors, 2016, 9(1):146. DOI:10.1186/s13071-016-1423-7.
[7] Li H, Cui XM, Cui N, et al. Human infection with novel spotted fever group Rickettsia genotype, China, 2015[J]. Emerg Infect Dis, 2016, 22(12):2153-2156. DOI:10.3201/eid2212.160962.
[8] 王卓, 王建伟, 于淼, 等. 东北地区蜱传斑点热群立克次体的分子流行病学研究[J]. 中国媒介生物学及控制杂志, 2018, 29(4):344-347. DOI:10.11853/j.issn.1003.8280.2018.04.005. Wang Z, Wang JW, Yu M, et al. Molecular epidemiological studies on spotted fever group Rickettsia in ticks from Northeastern China[J]. Chin J Vector Biol Control, 2018, 29(4):344-347. DOI:10.11853/j.issn.1003.8280.2018.04.005.(in Chinese)
[9] 张艳凯, 刘敬泽. 蜱类病原体和共生菌多样性及其作用[J]. 应用昆虫学报, 2019, 56(1):1-11. DOI:10.7679/j.issn.2095-1353.2019.001. Zhang YK, Liu JZ. Diversity, and roles, of pathogens and symbionts in ticks[J]. Chin J Appl Entomol, 2019, 56(1):1-11. DOI:10.7679/j.issn.2095-1353.2019.001.(in Chinese)
[10] 周磊, 汤芳, 栾进, 等. 内蒙古奇乾地区蜱携带斑点热立克次体的调查[J]. 中国国境卫生检疫杂志, 2017, 40(2):96-99. DOI:10.16408/j.1004-9770.2017.02.006. Zhou L, Tang F, Luan J, et al. Investigation on spotted fever group rickettsiae in ticks collected from Qiqian areas of Inner Mongolia[J]. Chin J Front Health Quarant, 2017, 40(2):96-99. DOI:10.16408/j.1004-9770.2017.02.006.(in Chinese)
[11] 冯立, 王卓, 杨军, 等. 黑龙江逊克地区森林革蜱斑点热立克次体DNA的检测[J]. 中国人兽共患病学报, 2014, 30(10):1020-1023. DOI:10.3969/cjz.j.issn.1002-2694.2014.10.007. Feng L, Wang Z, Yang J, et al. Molecular detection of spotted fever group Rickettsia in Dermacentor silvarum from the Xunke area, China[J]. Chin J Zoonoses, 2014, 30(10):1020-1023. DOI:10.3969/cjz.j.issn.1002-2694.2014.10.007.(in Chinese)
[12] Parola P, Paddock CD, Raoult D. Tick-borne rickettsioses around the world:Emerging diseases challenging old concepts[J]. Clin Microbiol Rev, 2005, 18(4):719-756. DOI:10.1128/CMR.18. 4.719-756.2005.
[13] 于志军, 刘敬泽. 蜱传疾病及其媒介蜱类研究进展[J]. 应用昆虫学报, 2015, 52(5):1072-1087. DOI:10.7679/j.issn.2095-1353.2015.128. Yu ZJ, Liu JZ. Progress in research on tick-borne diseases and vector ticks[J]. Chin J Appl Entomol, 2015, 52(5):1072-1087. DOI:10.7679/j.issn.2095-1353.2015.128.(in Chinese)
[14] 李思思, 张晓雨, 张艳凯, 等. 内蒙古地区全沟硬蜱细菌群落结构及多样性分析[J]. 中国媒介生物学及控制杂志, 2019, 30(6):607-612. DOI:10.11853/j.issn.1003.8280.2019.06.003. Li SS, Zhang XY, Zhang YK, et al. An analysis of bacterial community composition and diversity in Ixodes persulcatus in Inner Mongolia, China[J]. Chin J Vector Biol Control, 2019, 30(6):607-612. DOI:10.11853/j.issn.1003.8280.2019.06.003.(in Chinese)
[15] 高杨, 许士奇, 李丽静, 等. 塞罕坝自然保护区硬蜱调查及分子鉴定[J]. 中国病原生物学杂志, 2019, 14(9):1054-1057, 1075. DOI:10.13350/j.cjpb.190914. Gao Y, Xu SQ, Li LJ, et al. Molecular identification of ixodid ticks collected from Saihanba in Hebei province[J]. J Parasit Biol, 2019, 14(9):1054-1057, 1075. DOI:10.13350/j.cjpb. 190914.(in Chinese)
[16] 周明浩, 陈红娜. 我国新发蜱媒病原体研究概述[J]. 中华卫生杀虫药械, 2019, 25(3):193-198. DOI:10.19821/j.1671-2781.2019.03.001. Zhou MH, Chen HN. A review on emerging tick-borne pathogens in China[J]. Chin J Hyg Insect Equip, 2019, 25(3):193-198. DOI:10.19821/j.1671-2781.2019.03.001.(in Chinese)
[17] 乌兰图雅, 殷旭红, 崔云虹, 等. 内蒙古中西部草原蜱媒病原体多样性及基因型分析[J]. 中国寄生虫学与寄生虫病杂志, 2021, 39(1):27-34, 42. DOI:10.12140/j.issn.1000-7423.2021. 01.004. Wulantuya, Yin XH, Cui YH, et al. Diversity and genotype analysis of tick-borne pathogens in grasslands in the central and western part of Inner Mongolia[J]. Chin J Parasitol Parasit Dis, 2021, 39(1):27-34, 42. DOI:10.12140/j.issn.1000-7423.2021. 01.004.(in Chinese)
[18] 闻静, 焦丹, 鞠文东, 等. 新发蜱传病原体:劳氏立克次体的研究现状[J]. 中国人兽共患病学报, 2015, 31(4):361-364. DOI:10.3969/cjz.j.issn.1002-2694.2015.04.015. Wen J, Jiao D, Ju WD, et al. Emerging tick-borne pathogen:The research status on Rickettsia raoultii[J]. Chin J Zoonoses, 2015, 31(4):361-364. DOI:10.3969/cjz.j.issn.1002-2694.2015.04. 015.(in Chinese)
[19] 桂峥, 于晶峰, 木兰. 内蒙古部分地区草原革蜱携带斑点热群立克次体DNA检测及其基因型分布[J]. 吉林大学学报(医学版), 2021, 47(1):210-215. DOI:10.13481/j.1671-587Ⅹ. 20210129. Gui Z, Yu JF, Mu L. Detection of DNA in spotted fever group Rickettsia carried by Dermacentor nuttalli in partial areas of Inner Mongolia and its distribution of genotypes[J]. J Jilin Univ(Med Ed)2021, 47(1):210-215. DOI:10.13481/j.1671⁃587Ⅹ. 20210129. (in Chinese)
[20] 姜理平, 孟真, 崔清荣, 等. 浙江省蜱标本中斑点热群立克次体rOmpA和gltA基因检测[J]. 中国媒介生物学及控制杂志, 2010, 21(4):350-352. Jiang LP, Meng Z, Cui QR, et al. Detection of rOmpA and gltA genes of spotted fever group rickettsiae from tick specimens in Zhejiang province[J]. Chin J Vector Biol Control, 2010, 21(4):350-352. (in Chinese)
[21] 徐广, 方庆权, Keirans JE, et al. 分子系统进化关系分析的一种新方法:贝叶斯法在硬蜱属中的应用[J]. 动物学报, 2003, 49(3):380-388. DOI:10.3969/j.issn.1674-5507.2003.03.013. Xu G, Fang QQ, Keirans JE, et al. Application of Bayesian method for inferring phylogenetic relationships of ticks within the genus Ixodes[J]. Acta Zool Sin, 2003, 49(3):380-388. DOI:10.3969/j.issn.1674-5507.2003.03.013.(in Chinese)