目的 对埃及伊蚊6个丝氨酸蛋白酶(CLIPs)基因进行系统分析,为埃及伊蚊先天免疫功能的研究提供理论基础。方法 利用生物信息学研究方法,使用在线软件Clustal Omega分析CLIPs在物种间的相似性,利用在线工具SOPMA对其二级结构进行预测,利用MEGA 7.0软件构建系统进化树;通过实时荧光定量PCR(RT-qPCR)方法研究6个CLIPs基因在埃及伊蚊从卵到成虫的发育时期及未吸血雌蚊不同组织中的表达情况;通过细菌侵染雌性成蚊,研究6个CLIPs对不同细菌感染时的诱导动力学。采用单因素方差分析对实验结果进行比较。结果 从美国国立生物技术信息中心的GeneBank数据库中得到埃及伊蚊6个CLIPs基因,通过生物信息学分析,发现其氨基酸序列在羧基端蛋白酶结构域同源性较高,在蛋白的二级结构中,无规卷曲所占比重最高,占53.82%~62.50%。系统发育分析表明,埃及伊蚊6个CLIPs基因存在2个不同的分支,其中1个分支与冈比亚按蚊2个CLIPs基因有较高的同源性,另1个分支与黑腹果蝇SP5有较高的同源性。时空表达谱结果显示,埃及伊蚊6个CLIPs基因在4龄期幼虫和成蚊中表达水平较为丰富,在蛹期表达水平均较低;在雌性成蚊不同组织中表达水平不同,胸部和脂肪体中均有较高的表达水平,而在马氏管、中肠和卵巢中的表达水平均较低。在细菌感染方面,埃及伊蚊6个CLIPs有着不同的诱导动力学,存在应对微生物侵染时的特异性反应。结论 在埃及伊蚊6个CLIPs蛋白序列中,无规卷曲所占比重最高,在进化关系上,与其他昆虫的CLIPs有较高的同源性。时空表达谱显示,6个CLIPs在伊蚊发育过程和成蚊各个组织中有着不同的表达模式,在应对不同细菌侵染时有着不同的诱导动力学。
Objective To systematically analyze six clip-domain serine protease (CLIPs) genes in Aedes aegypti, so as to provide a theoretical basis for the study of their innate immune function in Ae. aegypti. Methods The bioinformatics software Clustal Omega was used to analyze the similarity of CLIPs among species, SOPMA was used to predict the secondary structure of CLIPs, and MEGA 7.0 software was used to construct a phylogenetic tree. RT-qPCR was performed to study the expression levels of CLIPs genes in Ae. aegypti from egg to adult and in different tissues of non-blood-fed adult female mosquitoes. The kinetics of immunity induction by CLIPs against infection with different bacterial species was studied by infecting female adult mosquitoes with bacteria. One-way ANOVA was used for statistical comparisons.Results Six CLIPs genes of Ae. aegypti were obtained from GenBank at NCBI. Bioinformatics analysis showed high amino acid sequence similarity in the carboxyl terminal of the six CLIPs genes. Random coils accounted for highest proportion (53.82%-62.50%) in the secondary structure of these proteins. The phylogenetic analysis showed that the six CLIPs genes were divided into two branches, one had high homology with two CLIPs genes in Anopheles gambiae, and the other branch had high homology with the SP5 of Drosophila melanogaster. The results of temporal and spatial expression profiles showed that the expression levels of the six CLIPs genes in Ae. aegypti were relatively high in the 4th instar larva and adult mosquitoes but low in pupal stage. The expression levels were different in different tissues of female adult mosquitoes, with high expression in thorax and fat body but low expression in malpighian tubule, midgut, and ovary. In terms of bacterial infection, the six CLIPs had different induction kinetics, suggesting that CLIPs in Ae. aegypti have specific response to microbial infection. Conclusion Random coils account for the highest proportion in the six CLIPs of Ae. aegypti. The six CLIPs have high homology with the CLIPs in other insect species. The spatiotemporal expression profiles show that the six CLIPs have different expression patterns during the development of Ae. aegypti and in different tissues of adult mosquitoes. They have different induction kinetics in response to infection with different bacterial species.
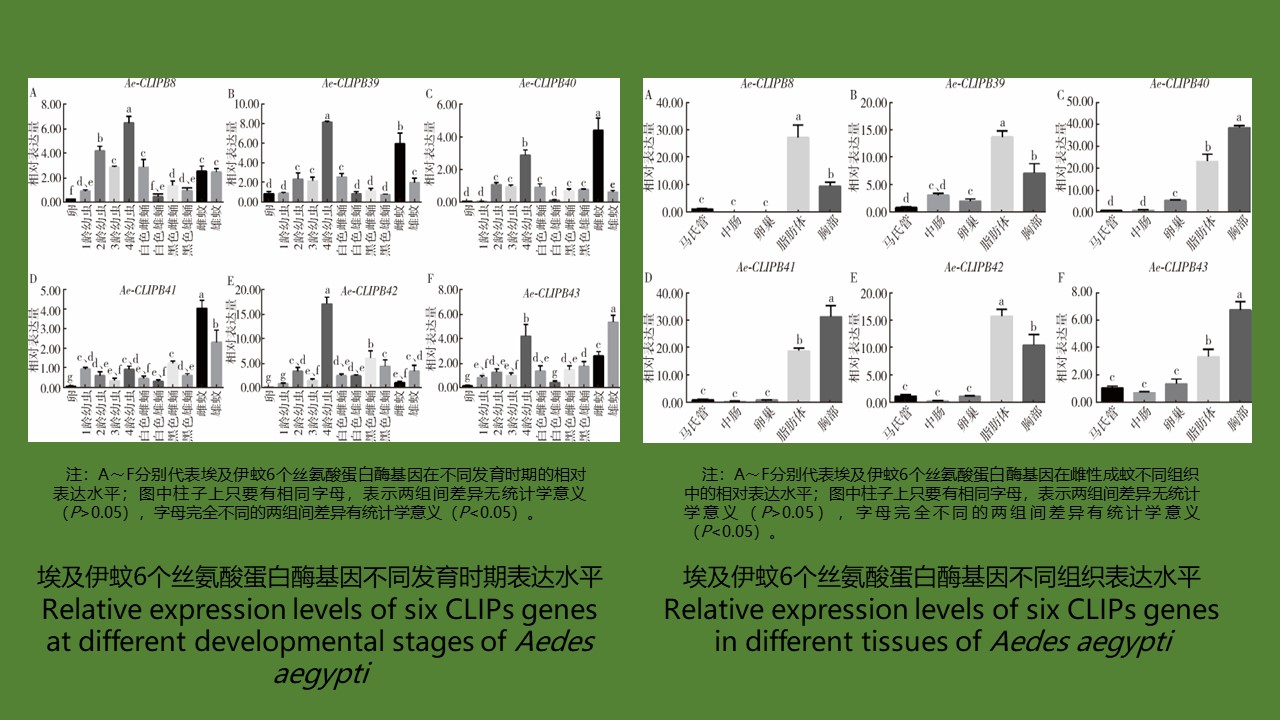
[1] Waterhouse RM, Kriventseva EV, Meister S, et al. Evolutionary dynamics of immune-related genes and pathways in disease-vector mosquitoes[J]. Science, 2007, 316(5832):1738-1743. DOI:10.1126/science.1139862.
[2] Liu HW, Heng JY, Wang LL, et al. Identification, characterization, and expression analysis of clip-domain serine protease genes in the silkworm, Bombyx mori[J]. Dev Comp Immunol, 2020, 105:103584. DOI:10.1016/j.dci.2019.103584.
[3] Gulley MM, Zhang X, Michel K. The roles of serpins in mosquito immunology and physiology[J]. J Insect Physiol, 2013, 59(2):138-147. DOI:10.1016/j.jinsphys.2012.08.015.
[4] Gubb D, Sanz-Parra A, Barcena L, et al. Protease inhibitors and proteolytic signalling cascades in insects[J]. Biochimie, 2010, 92(12):1749-1759. DOI:10.1016/j.biochi.2010.09.004.
[5] Park JW, Kim CH, Rui J, et al. Beetle immunity[J]. Adv Exp Med Biol, 2010, 708:163-180. DOI:10.1007/978-1-4419-8059-5_9.
[6] Jiang HB, Vilcinskas A, Kanost MR. Immunity in lepidopteran insects[J]. Adv Exp Med Biol, 2010, 708:181-204. DOI:10.1007/978-1-4419-8059-5_10.
[7] Rajah MM, Pardy RD, Condotta SA, et al. Zika virus:Emergence, phylogenetics, challenges, and opportunities[J]. ACS Infect Dis, 2016, 2(11):763-772. DOI:10.1021/acsinfecdis.6b00161.
[8] Zou Z, Shin SW, Alvarez KS, et al. Distinct melanization pathways in the mosquito Aedes aegypti[J]. Immunity, 2010, 32(1):41-53. DOI:10.1016/j.immuni.2009.11.011.
[9] Ballester M, Cordr M, CFolch JM. DAG expression:high-throughput gene expression analysis of real-time PCR data using standard curves for relative quantification[J]. PLoS One, 2013, 8(11):e80385. DOI:10.1371/journal.pone.0080385.
[10] Madeira F, Park YM, Lee J, et al. The EMBL-EBI search and sequence analysis tools APIs in 2019[J]. Nucleic Acids Res, 2019, 47(W1):W636-641. DOI:10.1093/nar/gkz268.
[11] Di Cera E. Serine proteases[J]. IUBMB Life, 2009, 61(5):510-515. DOI:10.1002/iub.186.
[12] Perona JJ, Craik CS. Structural basis of substrate specificity in the serine proteases[J]. Protein Sci, 1995, 4(3):337-360. DOI:10.1002/pro.5560040301.
[13] De Gregorio E, Han SJ, Lee WJ, et al. An immune-responsive serpin regulates the melanization cascade in Drosophila[J]. Dev Cell, 2002, 3(4):581-592. DOI:10.1016/S1534-5807(02)00267-8.
[14] Ligoxygakis P, Pelte N, Ji CY, et al. A serpin mutant links Toll activation to melanization in the host defence of Drosophila[J]. EMBO J, 2002, 21(23):6330-6337. DOI:10.1093/emboj/cdf661.
[15] Dohke K. Studies on prephenoloxidase-activating enzyme from cuticle of the silkworm Bombyx mori:I. Activation reaction by the enzyme[J]. Arch Biochem Biophys, 1973, 157(1):203-209. DOI:10.1016/0003-9861(73)90406-2.
[16] Dohke K. Studies on prephenoloxidase-activating enzyme from cuticle of the silkworm Bombyx mori:II. Purification and characterization of the enzyme[J]. Arch Biochem Biophys, 1973, 157(1):210-221. DOI:10.1016/0003-9861(73)90407-4.
[17] Barillas-Mury C. CLIP proteases and Plasmodium melanization in Anopheles gambiae[J]. Trends Parasitol, 2007, 23(7):297-299. DOI:10.1016/j.pt.2007.05.001.
[18] Volz J, Osta MA, Kafatos FC, et al. The roles of two clip domain serine proteases in innate immune responses of the malaria vector Anopheles gambiae[J]. J Biol Chem, 2005, 280(48):40161-40168. DOI:10.1074/jbc.M506191200.
[19] Wang RG, Lee SY, Cerenius L, et al. Properties of the prophenoloxidase activating enzyme of the freshwater crayfish, Pacifastacus leniusculus[J]. Eur J Biochem, 2001, 268(4):895-902. DOI:10.1046/j.1432-1327.2001.01945.x.
[20] Lemaitre B, Hoffmann J. The host defense of Drosophila melanogaster[J]. Ann Rev Immunol, 2007, 25:697-743. DOI:10.1146/annurev.immunol.25.022106.141615.
[21] Xi ZY, Ramirez JL, Dimopoulos G. The Aedes aegypti toll pathway controls dengue virus infection[J]. PLoS Pathog, 2008, 4(7):e1000098. DOI:10.1371/journal.ppat.1000098.
[22] Wang HC, Wang QH, Bhowmick B, et al. Functional characterization of two clip domain serine proteases in innate immune responses of Aedes aegypti[J]. Parasit Vectors, 2021, 14(1):584. DOI:10.1186/s13071-021-05091-9.
[23] Bai L, Wang LL, Vega-Rodríguez J, et al. A gut symbiotic bacterium Serratia marcescens renders mosquito resistance to Plasmodium infection through activation of mosquito immune responses[J]. Front Microbiol, 2019, 10:1580. DOI:10.3389/fmicb.2019.01580.
[24] Sim S, Jupatanakul N, Dimopoulos G. Mosquito immunity against arboviruses[J]. Viruses, 2014, 6(11):4479-4504. DOI:10.3390/v6114479.
[25] Castillejo-Lspez C, Häcker U. The serine protease Sp7 is expressed in blood cells and regulates the melanization reaction in Drosophila[J]. Biochem Biophys Res Commun, 2005, 338(2):1075-1082. DOI:10.1016/j.bbrc.2005.10.042.



