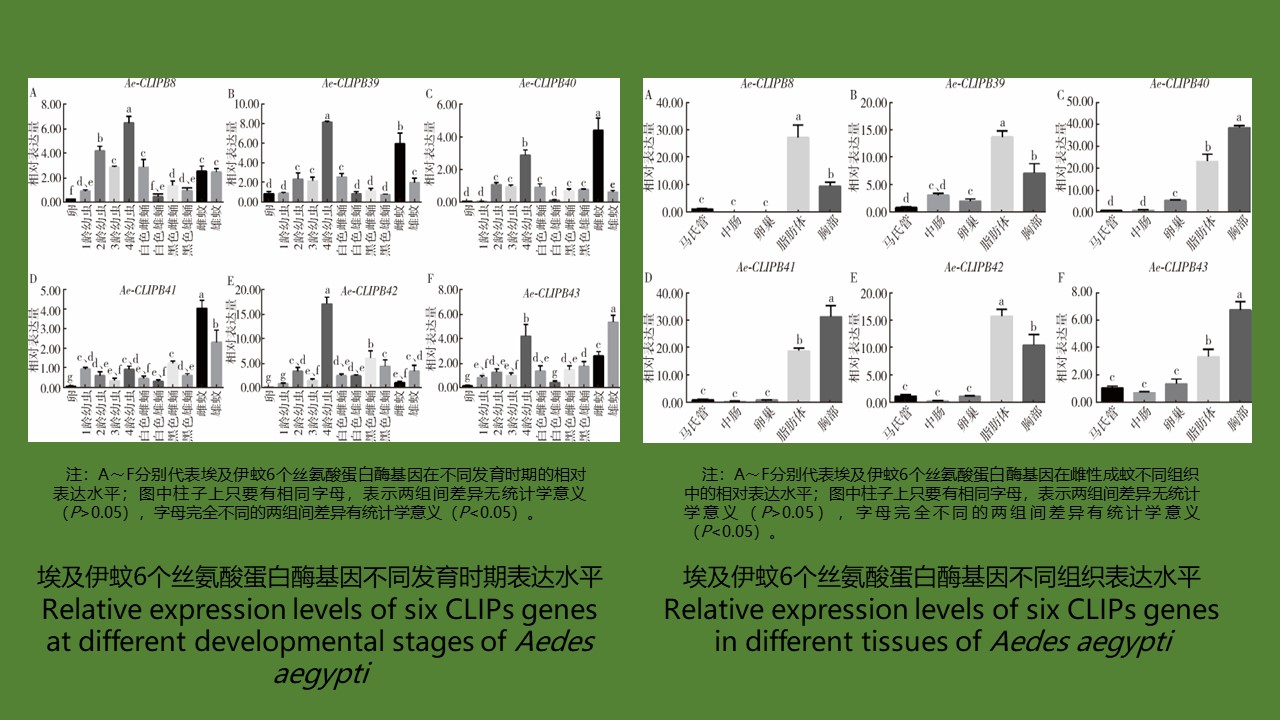
Objective To systematically analyze six clip-domain serine protease (CLIPs) genes in Aedes aegypti, so as to provide a theoretical basis for the study of their innate immune function in Ae. aegypti. Methods The bioinformatics software Clustal Omega was used to analyze the similarity of CLIPs among species, SOPMA was used to predict the secondary structure of CLIPs, and MEGA 7.0 software was used to construct a phylogenetic tree. RT-qPCR was performed to study the expression levels of CLIPs genes in Ae. aegypti from egg to adult and in different tissues of non-blood-fed adult female mosquitoes. The kinetics of immunity induction by CLIPs against infection with different bacterial species was studied by infecting female adult mosquitoes with bacteria. One-way ANOVA was used for statistical comparisons.Results Six CLIPs genes of Ae. aegypti were obtained from GenBank at NCBI. Bioinformatics analysis showed high amino acid sequence similarity in the carboxyl terminal of the six CLIPs genes. Random coils accounted for highest proportion (53.82%-62.50%) in the secondary structure of these proteins. The phylogenetic analysis showed that the six CLIPs genes were divided into two branches, one had high homology with two CLIPs genes in Anopheles gambiae, and the other branch had high homology with the SP5 of Drosophila melanogaster. The results of temporal and spatial expression profiles showed that the expression levels of the six CLIPs genes in Ae. aegypti were relatively high in the 4th instar larva and adult mosquitoes but low in pupal stage. The expression levels were different in different tissues of female adult mosquitoes, with high expression in thorax and fat body but low expression in malpighian tubule, midgut, and ovary. In terms of bacterial infection, the six CLIPs had different induction kinetics, suggesting that CLIPs in Ae. aegypti have specific response to microbial infection. Conclusion Random coils account for the highest proportion in the six CLIPs of Ae. aegypti. The six CLIPs have high homology with the CLIPs in other insect species. The spatiotemporal expression profiles show that the six CLIPs have different expression patterns during the development of Ae. aegypti and in different tissues of adult mosquitoes. They have different induction kinetics in response to infection with different bacterial species.
WANG Qiu-hui, WANG Hao-cheng, HAN Qian, LIAO Cheng-hong
. Bioinformatics analysis and preliminary functional study of six CLIPs genes in Aedes aegypti[J]. Chinese Journal of Vector Biology and Control, 2022
, 33(2)
: 183
-190
.
DOI: 10.11853/j.issn.1003.8280.2022.02.004
[1] Waterhouse RM, Kriventseva EV, Meister S, et al. Evolutionary dynamics of immune-related genes and pathways in disease-vector mosquitoes[J]. Science, 2007, 316(5832):1738-1743. DOI:10.1126/science.1139862.
[2] Liu HW, Heng JY, Wang LL, et al. Identification, characterization, and expression analysis of clip-domain serine protease genes in the silkworm, Bombyx mori[J]. Dev Comp Immunol, 2020, 105:103584. DOI:10.1016/j.dci.2019.103584.
[3] Gulley MM, Zhang X, Michel K. The roles of serpins in mosquito immunology and physiology[J]. J Insect Physiol, 2013, 59(2):138-147. DOI:10.1016/j.jinsphys.2012.08.015.
[4] Gubb D, Sanz-Parra A, Barcena L, et al. Protease inhibitors and proteolytic signalling cascades in insects[J]. Biochimie, 2010, 92(12):1749-1759. DOI:10.1016/j.biochi.2010.09.004.
[5] Park JW, Kim CH, Rui J, et al. Beetle immunity[J]. Adv Exp Med Biol, 2010, 708:163-180. DOI:10.1007/978-1-4419-8059-5_9.
[6] Jiang HB, Vilcinskas A, Kanost MR. Immunity in lepidopteran insects[J]. Adv Exp Med Biol, 2010, 708:181-204. DOI:10.1007/978-1-4419-8059-5_10.
[7] Rajah MM, Pardy RD, Condotta SA, et al. Zika virus:Emergence, phylogenetics, challenges, and opportunities[J]. ACS Infect Dis, 2016, 2(11):763-772. DOI:10.1021/acsinfecdis.6b00161.
[8] Zou Z, Shin SW, Alvarez KS, et al. Distinct melanization pathways in the mosquito Aedes aegypti[J]. Immunity, 2010, 32(1):41-53. DOI:10.1016/j.immuni.2009.11.011.
[9] Ballester M, Cordr M, CFolch JM. DAG expression:high-throughput gene expression analysis of real-time PCR data using standard curves for relative quantification[J]. PLoS One, 2013, 8(11):e80385. DOI:10.1371/journal.pone.0080385.
[10] Madeira F, Park YM, Lee J, et al. The EMBL-EBI search and sequence analysis tools APIs in 2019[J]. Nucleic Acids Res, 2019, 47(W1):W636-641. DOI:10.1093/nar/gkz268.
[11] Di Cera E. Serine proteases[J]. IUBMB Life, 2009, 61(5):510-515. DOI:10.1002/iub.186.
[12] Perona JJ, Craik CS. Structural basis of substrate specificity in the serine proteases[J]. Protein Sci, 1995, 4(3):337-360. DOI:10.1002/pro.5560040301.
[13] De Gregorio E, Han SJ, Lee WJ, et al. An immune-responsive serpin regulates the melanization cascade in Drosophila[J]. Dev Cell, 2002, 3(4):581-592. DOI:10.1016/S1534-5807(02)00267-8.
[14] Ligoxygakis P, Pelte N, Ji CY, et al. A serpin mutant links Toll activation to melanization in the host defence of Drosophila[J]. EMBO J, 2002, 21(23):6330-6337. DOI:10.1093/emboj/cdf661.
[15] Dohke K. Studies on prephenoloxidase-activating enzyme from cuticle of the silkworm Bombyx mori:I. Activation reaction by the enzyme[J]. Arch Biochem Biophys, 1973, 157(1):203-209. DOI:10.1016/0003-9861(73)90406-2.
[16] Dohke K. Studies on prephenoloxidase-activating enzyme from cuticle of the silkworm Bombyx mori:II. Purification and characterization of the enzyme[J]. Arch Biochem Biophys, 1973, 157(1):210-221. DOI:10.1016/0003-9861(73)90407-4.
[17] Barillas-Mury C. CLIP proteases and Plasmodium melanization in Anopheles gambiae[J]. Trends Parasitol, 2007, 23(7):297-299. DOI:10.1016/j.pt.2007.05.001.
[18] Volz J, Osta MA, Kafatos FC, et al. The roles of two clip domain serine proteases in innate immune responses of the malaria vector Anopheles gambiae[J]. J Biol Chem, 2005, 280(48):40161-40168. DOI:10.1074/jbc.M506191200.
[19] Wang RG, Lee SY, Cerenius L, et al. Properties of the prophenoloxidase activating enzyme of the freshwater crayfish, Pacifastacus leniusculus[J]. Eur J Biochem, 2001, 268(4):895-902. DOI:10.1046/j.1432-1327.2001.01945.x.
[20] Lemaitre B, Hoffmann J. The host defense of Drosophila melanogaster[J]. Ann Rev Immunol, 2007, 25:697-743. DOI:10.1146/annurev.immunol.25.022106.141615.
[21] Xi ZY, Ramirez JL, Dimopoulos G. The Aedes aegypti toll pathway controls dengue virus infection[J]. PLoS Pathog, 2008, 4(7):e1000098. DOI:10.1371/journal.ppat.1000098.
[22] Wang HC, Wang QH, Bhowmick B, et al. Functional characterization of two clip domain serine proteases in innate immune responses of Aedes aegypti[J]. Parasit Vectors, 2021, 14(1):584. DOI:10.1186/s13071-021-05091-9.
[23] Bai L, Wang LL, Vega-Rodríguez J, et al. A gut symbiotic bacterium Serratia marcescens renders mosquito resistance to Plasmodium infection through activation of mosquito immune responses[J]. Front Microbiol, 2019, 10:1580. DOI:10.3389/fmicb.2019.01580.
[24] Sim S, Jupatanakul N, Dimopoulos G. Mosquito immunity against arboviruses[J]. Viruses, 2014, 6(11):4479-4504. DOI:10.3390/v6114479.
[25] Castillejo-Lspez C, Häcker U. The serine protease Sp7 is expressed in blood cells and regulates the melanization reaction in Drosophila[J]. Biochem Biophys Res Commun, 2005, 338(2):1075-1082. DOI:10.1016/j.bbrc.2005.10.042.


